UALR-UAMS partnership results in FDA-approved obstetric medical device
A partnership between researchers at UALR and the University of Arkansas for Medical Sciences has resulted in an inexpensive, disposable external medical device that monitors contractions in pregnant women. James D. Wilson, assistant director of research at the UALR Graduate Institute of Technology; Dr. Curtis Lowery, chairman of the UAMS College of Medicine’s Department of Obstetrics and Gynecology; and Hari Eswaran, Ph.D., a professor in the same department, created and developed the device, which was recently approved by the Food and Drug Administration.
UALR researcher, James D. Wilson, right, sits with Dr. Curtis Lowery, left, chairman of the UAMS College of Medicine’s Department of Obstetrics and Gynecology. The two collaborated with UAMS researcher Hari Eswaran, Ph.D., (in back) to help create an obstetric device recently approved by the Food and Drug Administration.
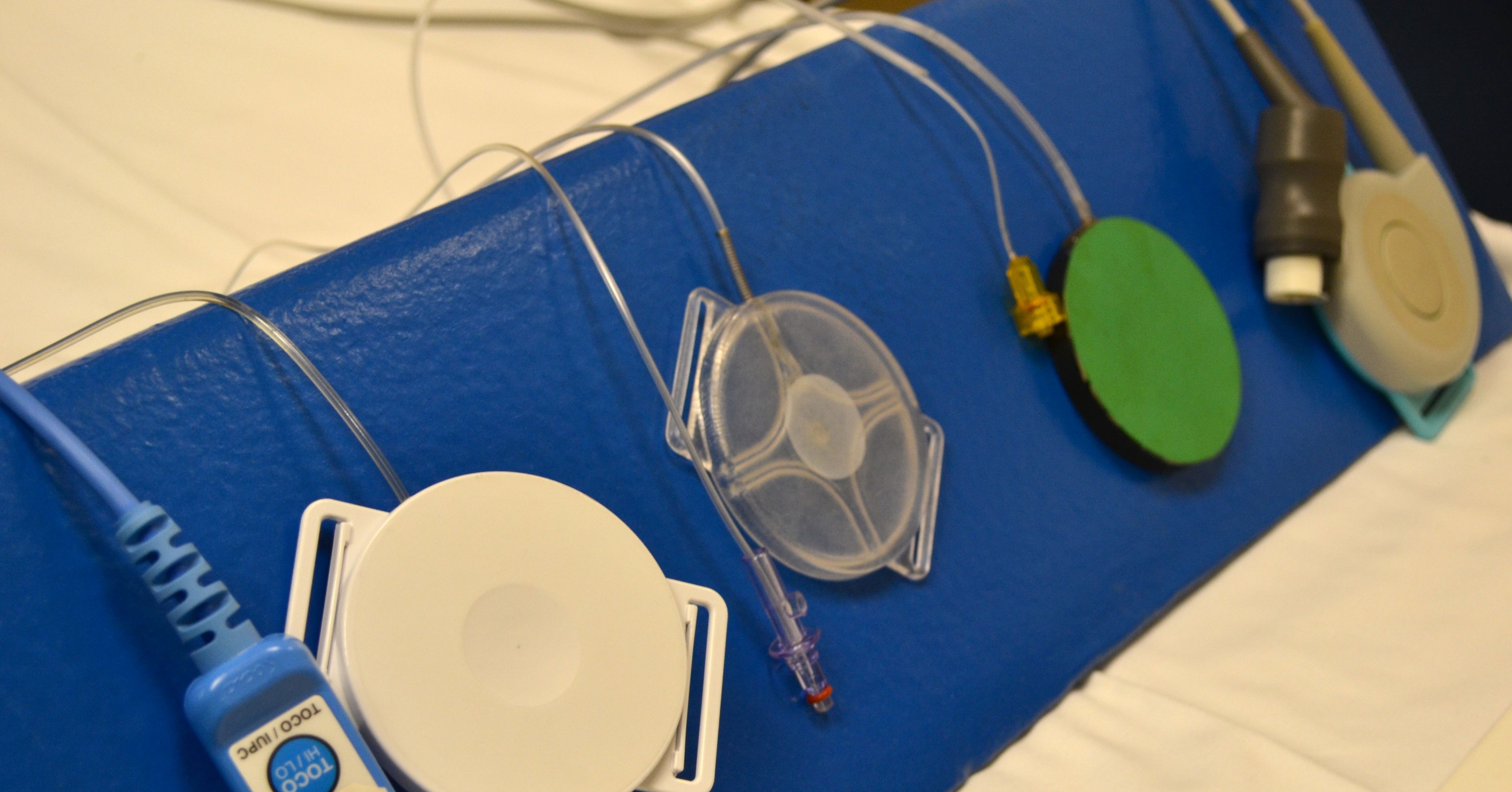 “That pushes the air inside, which produces a signal. The signal is transmitted through an attached cable that also is plugged into a fetal heart rate monitor,” said Wilson.
The older, electronic tocodynamometers are heavier and must be continually cleaned because perspiration, other bodily fluids, and medical gels come into contact with it, according to the researchers.
“Because Koala Toco costs about $15, the nurse doesn’t have to spend time later cleaning and sterilizing the device. Ours can be tossed out,” Wilson added.
Wilson most recently collaborated with Dr. William Culp, a professor of radiology, surgery, and neurology and vice chairman of research at UAMS, to develop the ClotBust ER®, which fits on the head like a halo to quickly bust clots that cause stroke.
“That pushes the air inside, which produces a signal. The signal is transmitted through an attached cable that also is plugged into a fetal heart rate monitor,” said Wilson.
The older, electronic tocodynamometers are heavier and must be continually cleaned because perspiration, other bodily fluids, and medical gels come into contact with it, according to the researchers.
“Because Koala Toco costs about $15, the nurse doesn’t have to spend time later cleaning and sterilizing the device. Ours can be tossed out,” Wilson added.
Wilson most recently collaborated with Dr. William Culp, a professor of radiology, surgery, and neurology and vice chairman of research at UAMS, to develop the ClotBust ER®, which fits on the head like a halo to quickly bust clots that cause stroke. View more stories in News
