Authors: Alokita Karmakar, Yang Xu, Thikra Mustafa, Ganesh Kannarpady, Stacie M. Bratton, Anna Radominska-Pandya, Peter A. Crooks, and Alexandru S Biris
Publication: RSC Advances, Issue 5, Issue 4
Abstract:
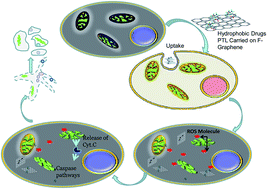
Advances in anticancer chemotherapy have been hindered by the lack of biocompatibility of new prospective drugs. One significant challenge concerns water insolubility, which compromises the bioavailability of the drugs leading to increased dosage and higher systemic toxicity. To overcome these problems, nanodelivery has been established as a promising approach for increasing the efficacy and lowering the required dosage of chemotherapeutics. The naturally derived compound, parthenolide (PTL), is known for its anti-inflammatory and anticancer activity, but its poor water solubility limits its clinical value. In the present study, we have used carboxyl-functionalized nanographene (fGn) delivery to overcome the extreme hydrophobicity of this drug. A water-soluble PTL analog, dimethylaminoparthenolide (DMAPT), was also examined for comparison with the anticancer efficacy of our PTL–fGn complex. Delivery by fGn was found to increase the anticancer/apoptotic effects of PTL (but not DMAPT) when delivered to the human pancreatic cancer cell line Panc-1. The IC50 value for PTL decreased from 39 μM to 9.5 μM when delivered as a mixture with fGn. The IC50 of DMAPT did not decrease when delivered as DMAPT–fGn and was significantly higher than that for PTL–fGn. There were significant increases in ROS formation and in mitochondrial membrane disruption in Panc-1 cells after PTL–fGn treatment as compared to PTL treatment, alone. Increases in toxicity were also seen with apoptosis detection assays using flow cytometry, ethidium bromide/acridine orange/DAPI staining, and TUNEL. Thus, fGn delivery was successfully used to overcome the poor water solubility of PTL, providing a strategy for improving the effectiveness of this anticancer agent.